
This probabilistic nature of the Schrödinger model allows for a more accurate description of atomic behavior, especially for complex systems with multiple electrons. The square of the wavefunction, known as the probability density, gives the likelihood of finding an electron in a specific region of space. These wavefunctions represent the probability distribution of finding an electron at a particular location. The Schrödinger equation, a fundamental equation in quantum mechanics, describes the behavior of electrons in terms of their wavefunctions. Unlike the Bohr model, the Schrödinger model does not provide a precise path or trajectory for electrons but rather describes their probable locations in three-dimensional space using mathematical equations called wavefunctions. This model introduced the concept of wave-particle duality, where electrons are described as both particles and waves. The Schrödinger model, also known as the quantum mechanical model, was developed by Erwin Schrödinger in the 1920s. The model failed to account for the repulsion between electrons and the resulting distortion of energy levels. However, it had limitations in explaining the behavior of atoms with more than one electron. It provided a framework for understanding the periodic table and the arrangement of elements based on their electron configurations. The Current Atomic Theory successfully explained the stability of atoms and the discrete nature of atomic spectra. Each energy level has a fixed energy value, and electrons can transition between these levels by absorbing or emitting energy in discrete packets called quanta. According to this theory, electrons orbit the nucleus in specific energy levels, much like planets orbiting the sun. This model revolutionized our understanding of atomic structure by introducing the concept of energy levels or shells. The Current Atomic Theory, also known as the Bohr model, was proposed by Niels Bohr in 1913. In this article, we will compare the attributes of these two theories and explore their implications in the realm of atomic physics. Two prominent theories that have shaped our understanding of atoms are the Current Atomic Theory and the Schrödinger model.
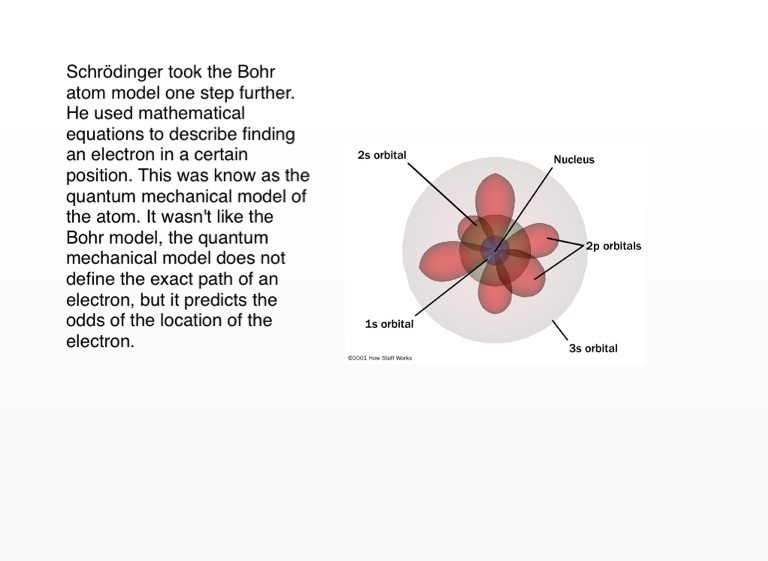
Over the years, our understanding of atomic theory has evolved, with significant contributions from various scientists. Atomic theory is a fundamental concept in the field of physics that seeks to explain the nature and behavior of matter at the atomic level.
